|
On the right-hand vertical axis, locate and mark the point containing the temperature 60☏.On the left-hand vertical axis, locate and mark the point containing the pressure 100 psia.Example įor example, to find the K value of methane at 100 psia and 60 ☏. 
Many DePriester charts have been printed for simple hydrocarbons. "K" values, representing the tendency of a given chemical species to partition itself preferentially between liquid and vapor phases, are plotted in between. 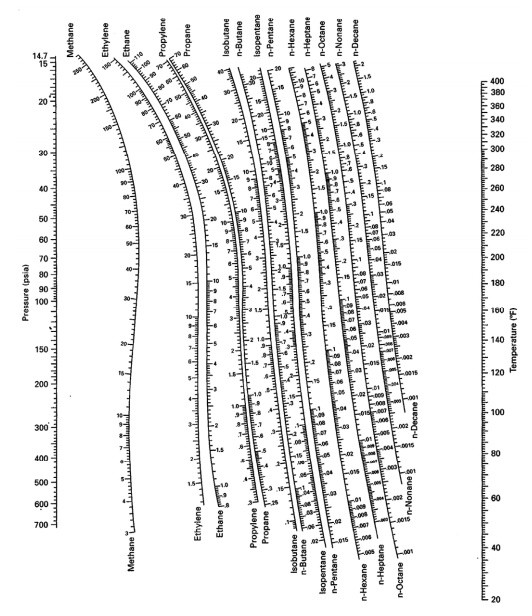
These nomograms have two vertical coordinates, one for pressure, and another for temperature. DePriester in an article in Chemical Engineering Progress in 1953. 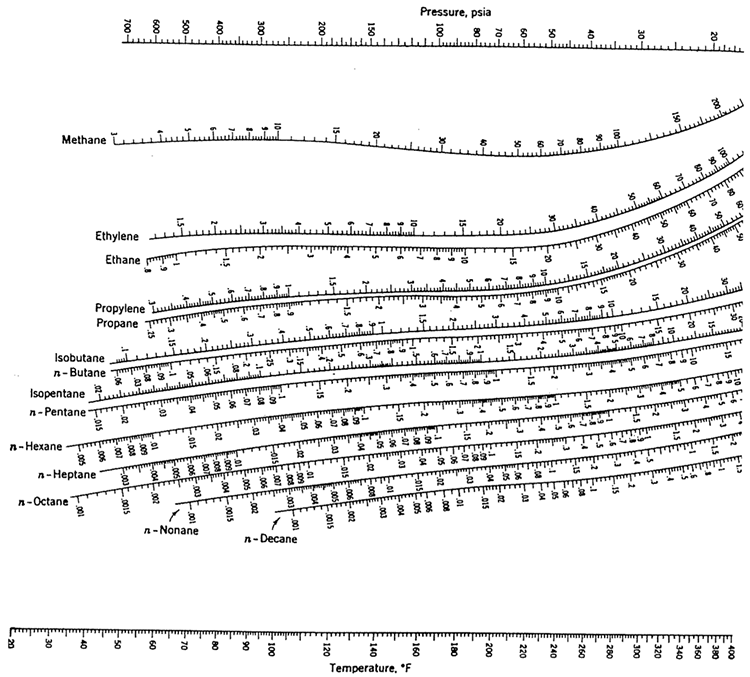
DePriester Charts provide an efficient method to find the vapor-liquid equilibrium ratios for different substances at different conditions of pressure and temperature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
